
Bacteria and fungi as well as polymicrobial organisms are known to form communities that adhere firmly to living or non-living surfaces in matrices of biofilms. These biofilm matrices are produced by cells into an extracellular polymeric substance (EPS), which is typically a conglomeration of extracellular polysaccharides, proteins, lipids and DNA. In the body, infectious biofilms can become antibiotic resistant and have become increasingly significant to the failure of medical implants and normal tissue repair. More than 80% of chronic wounds have bacteria, fungi or polymicrobial organisms living within a biofilm causing delayed wound healing.
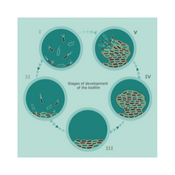
Biofilms evolve through different stages of development and reformation. They are initially seeded by free-floating microorganisms that begin to attach to surfaces. These are known as planktonic organisms. Planktonic bacteria as free-floating organisms serve to colonize new niches, and do not have the protection of biofilms making them easier to eradicate.
The attachment of these planktonic organisms is followed by a period of growth. During this growth the bacteria begin to multiply and become more firmly attached with changing gene expression patterns that support its survival. They begin to secrete a surrounding protective matrix known as the EPS. Additional layers of microorganisms and EPS development build upon the first attachment layers within several hours and are known as immature biofilm. Within 12 hours, the biofilm continues to evolve and mature, with the bacteria buried deeper into the matrix making it increasingly tolerant to biocides. This is known as mature biofilm and is very difficult to eradicate. Mature biofilm is well protected by its EPS and displays both antimicrobial tolerance, and immune response evasion. It also continuously sheds planktonic bacteria, microcolonies and fragments of biofilm, which can disperse and attach to other parts of the wound bed or to other wounds, forming new biofilm colonies.
The clinicians challenge to assessment of biofilm in a wound is that they are microscopic structures. They can’t be seen with the naked eye unless allowed to grow for an extended period of time in which they become thick and slimy in appearance. However, slough in a wound can look thick and slimy and may or may not have biofilm. Since there are no readily available means to diagnose biofilm at the bedside, suspicion of biofilm should be high in chronic wounds that fail to progress with good wound bed preparation and management.
Treating biofilms when strongly suspected requires a multifaceted approach. It incorporates physical removal through wound cleansing and debridement to disrupt the EPS. It then involves safe elimination of the bacteria with an antimicrobial agent that can target and kill the microbes and lastly, to prevent biofilm reformation with topical antimicrobial or barrier dressings.
